Your shopping cart is empty.

All orders will be shipped with Canada Post to the provided shipping address. Please allow 7-10 business days to receive your order.
Symbiopathic
In contemporary clinical practice, the integration of advanced and biologically attuned treatment modalities has become increasingly important. Symbiopathic remedies, grounded in the pioneering research of Dr. Günther Enderlein, offer a rigorously prepared, terrain-focused approach designed to support internal balance and biological regulation. Developed according to the principles of isopathy and homeopathy, these preparations provide a coherent framework for practitioners working within integrative and biological paradigms.
Symbiopathic remedies are precisely formulated homeopathic medicinal products that assist in modulating the internal milieu, fostering microbial symbiosis, and reinforcing the body's innate regulatory functions. Dr. Günther Enderlein, a distinguished German bacteriologist and immunologist, devoted his scientific career to exploring the dynamic interactions between microorganisms and human health. His foundational work continues to inform advanced biological and integrative medical models.
Our founder, a German-born expert trained in Switzerland, is one of only two individuals to have received direct instruction in Enderlein’s original methods from a senior protégé. He has preserved the fidelity of these preparations while guiding their ongoing refinement. Under his direction, Symbiopathic remedies continue to reflect the highest standards of the Enderlein tradition, combining historical precision with contemporary production technologies.
Through serial dilution and potentization, the bioactivity of each preparation is preserved and optimized while minimizing undesirable reactivity. Utilizing carefully selected fungal and bacterial strains, these remedies act upon foundational regulatory levels rather than targeting isolated symptoms. This systems-oriented approach considers the integrity of the internal environment as a whole.
Modern production methods ensure the exclusion of cell wall fragments, helping to minimize systemic burden while preserving the original terrain-regulating intent of the formulas. As such, Symbiopathic remedies are frequently incorporated into integrative clinical protocols that prioritize microbial balance, immune modulation, and long-term systemic support.
For practitioners, these remedies offer valuable tools in the management of complex and chronic presentations, especially within frameworks that address underlying dysregulation and terrain vulnerability. They may be integrated alongside conventional or complementary therapies to support the body’s broader biological coherence.
Symbiopathic – Biological Health Group Corp. represents a commitment to excellence in the field of homeopathic and biological medicine. With unwavering dedication to research integrity and pharmaceutical craftsmanship, Symbiopathic continues to support clinicians across the globe with high-quality formulations and consistent reliability. We are honored to serve those advancing integrative care in both established and emerging medical landscapes.
Homeopathic Remedies
Drug Facts acc. to Enderlein Symbio Albicans 5X Drops (10ml) (NDC 69710-195-11) Active Ingredient PurposeCandida albicans 5X...............Internal indications* Uses For temporary relief of minor irritation, itching or soreness associated with Candida albicans yeast infections. These statements are based upon traditional homeopathic practice. They have not been reviewed by the Food and Drug Administration. Warnings:If pregnant or...
(NDC 69710-255-10) Active Ingredient PurposeMycobacterium phlei 6X........... Internal indications* Uses This homeopathic remedy is indicated for symptoms associated with bacterial infections and inflammation according to traditional homeopathic practice. This includes those caused by staphylococci and streptococci. ✔ Chronic lung conditions✔ Pneumonia, TB, and autoimmune conditions✔ Stimulates lymphocyte (WBC) activity✔ Immune modulation with Mycobacterium phlei Indications: Symbio Myco Phlei...
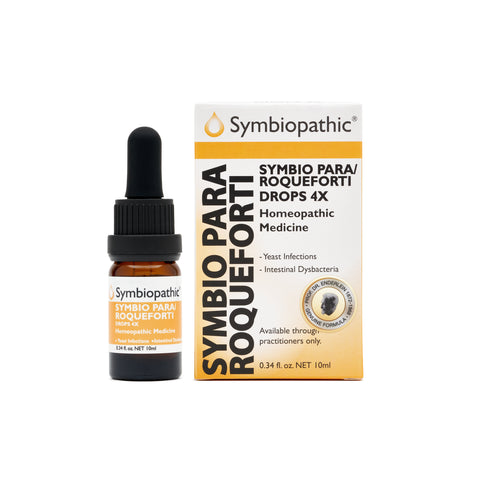
Drug Facts acc. to Enderlein Symbio Para-Roqueforti 4X Drops (10ml) (NDC 69710-165-11) Active Ingredient PurposeCandida parapsilosis 4X.......................Internal indications*Penicillium roqueforti 4X......................Internal indications* Uses This homeopathic remedy is indicated for symptoms associated with yeast infections and intestinal dysbacteria according to traditional homeopathic practice. * These statements...
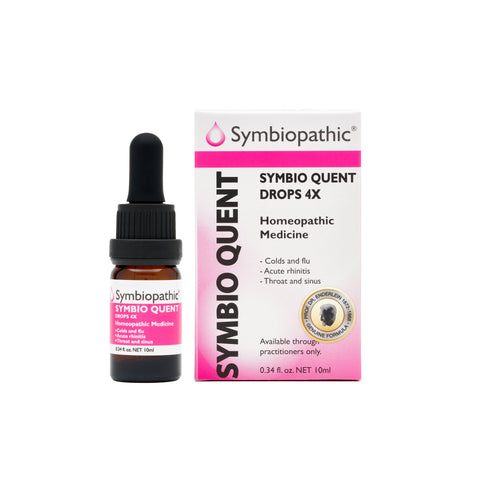
Drug Facts acc. to Enderlein Symbio Quent 4X Drops (10ml) (NDC 69710-175-11) Active Ingredient PurposePenicillium frequentans 4X..............Internal indications* Uses This homeopathic remedy is indicated for symptoms associated with colds; flu, inflamed nasal passages, pre- and post- influenza neuralgia, laryngitis, sinusitis and myalgias according to traditional homeopathic...
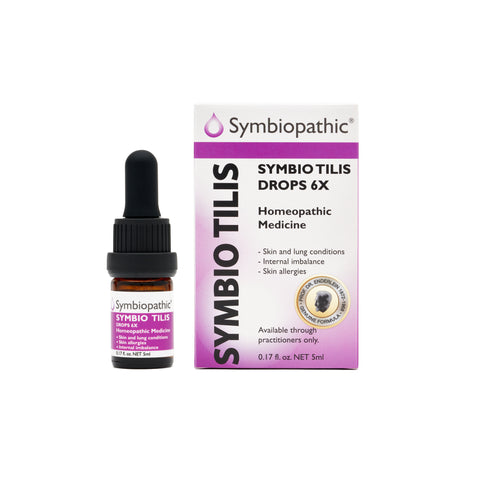
Drug Facts acc. to Enderlein (NDC 69710-235-11) Active Ingredient PurposeBacillus subtilis 6X..................Internal indications* Uses This remedy is indicated for symptoms associated with skin and lung conditions, stomach and intestinal imbalance, rheumatism symptoms, skin allergies, and other internal conditions according to traditional homeopathic practice. * These statements are based upon traditional homeopathic...
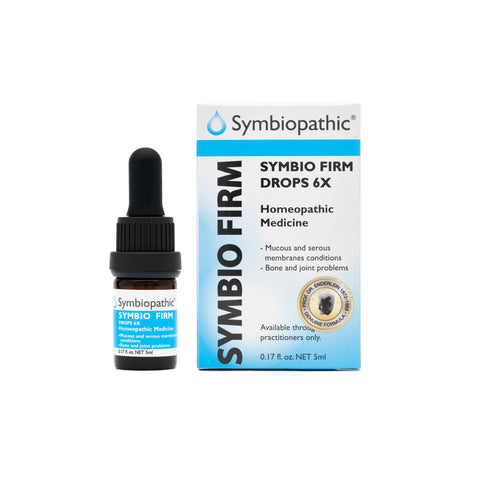
Drug Facts acc. to Enderlein (NDC 69710-215-11) Active Ingredient - PurposeBacillus firmus 6X......................Internal indications* Uses This remedy is indicated for symptoms associated with inflammation of serous and mucous membranes, bone infections, and joint problems according to traditional homeopathic practice. Indications: Firmus helps resolve chronic inflammations of serous and mucous membranes, bone infections, and joint problems. Indigenous to the human body,...
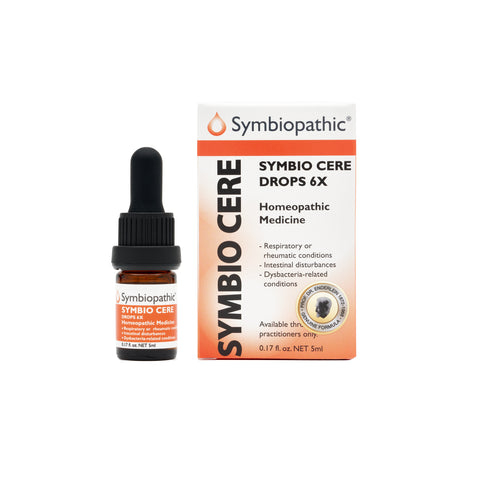
\Drug facts(NDC 69710-205-11) Active Ingredient - PurposeBacillus cereus 6X...........................Internal indications* Uses This remedy is indicated for symptoms associated with respiratory or rheumatic conditions, intestinal disturbances caused by dysbacteria, and other mucous membrane conditions according to traditional homeopathic practice. Indications: Cereus, a more potent strain of Bacillus subtilis, is indicated for the treatment of chronic respiratory or rheumatic inflammations, liver...
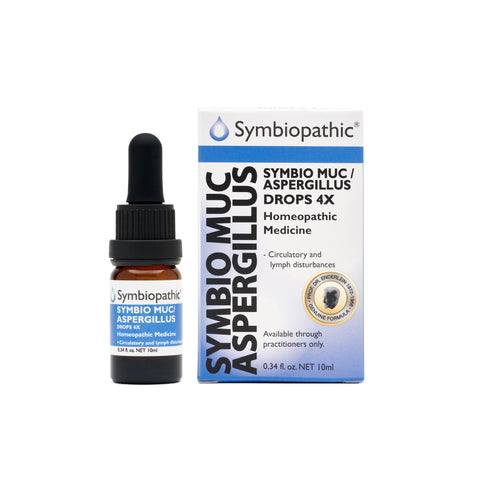
Drug Facts acc. to Enderlein Symbio Muc-Aspergillius 4X Drops (10ml) (NDC 69710-145-11) Active Ingredient PurposeMucor racemosus 4X....................Internal indications*Aspergillus niger 4X......................Internal indications* Uses This homeopathic remedy is indicated for symptoms associated with circulatory and lymph disturbances, including tonsillitis and chronic liver conditions according to traditional homeopathic practice. *These statements...
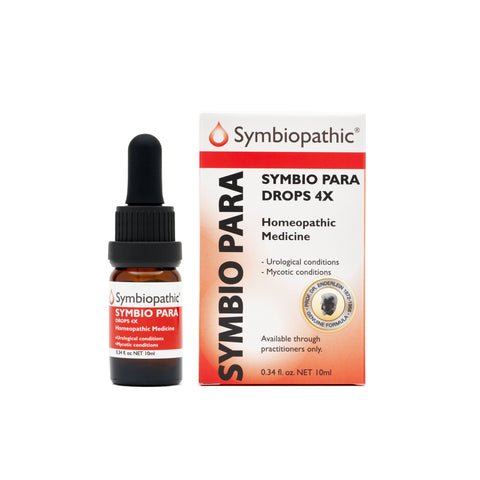
Drug Facts acc. to Enderlein Symbio Para 4X Drops (10ml) (NDC 69710-115-11) Active Ingredient PurposeCandida parapsilosis 4X...............Internal indications* Uses This homeopathic remedy is indicated for symptoms that afflict body orifices including mycotic infections, leucorrhea, dental foci, granuloma dentis, rhagades, and pernphigus according to traditional homeopathic practice. * These statements are based upon traditional homeopathic practice. They have not been reviewed...
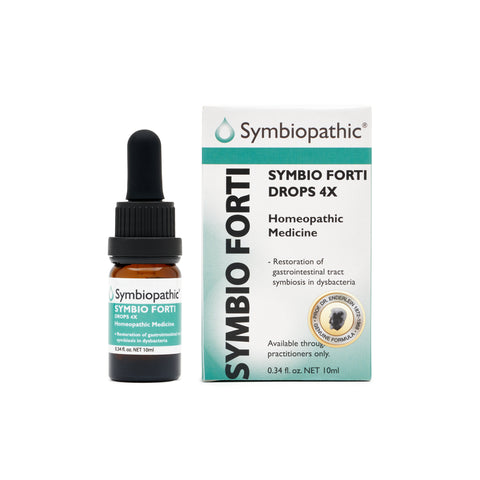
Drug Facts acc. to Enderlein Symbio Forti 4X Drops (10ml) (NDC 69710-185-11) Active Ingredient - PurposePenicillium roqueforti 4X................Internal indications* Uses This remedy is indicated for symptoms associated with gastrointestinal tract symbiosis restoration in cases of dysbacteria according to traditional homeopathic medicine practice. * These statements are based upon traditional homeopathic practice. They have not been reviewed by the Food and Drug...
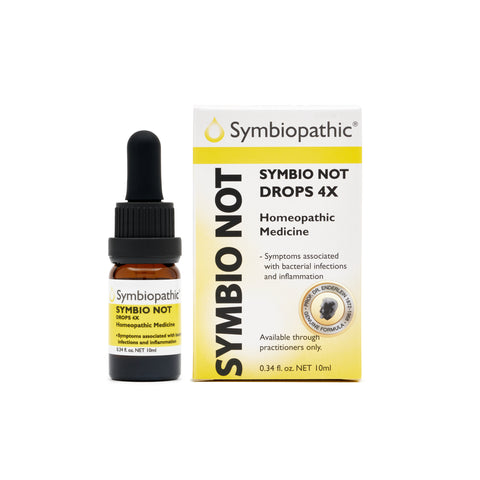
Drug Facts acc. to Enderlein Symbio Not 4X Drops (10ml) (NDC 69710-155-11) Active Ingredient PurposePenicillium notatum (chrysogenum) 4X..............Internal indications* Uses This remedy is indicated for symptoms associated with bacterial infections and inflammation according to traditional homeopathic medicine. This includes...
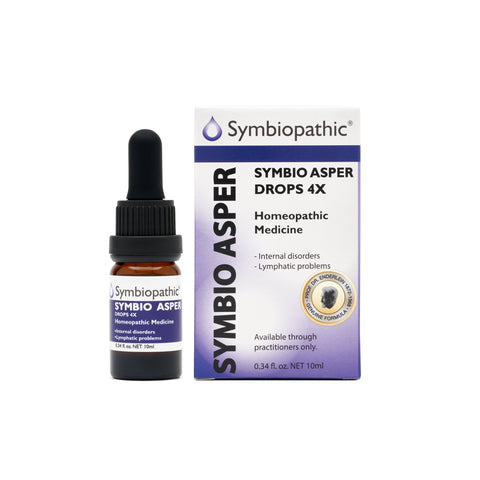
Drug Facts acc. to Enderlein Symbio Asper 4X Drops (10ml) (NDC 69710-105-11) Active Ingredient - PurposeAspergillus niger 4X........... Internal indications* Uses This homeopathic remedy is indicated for symptoms associated with allergies, rheumatic illnesses, stomach ulcers, hyperactive thyroid, lymphatic problems, and phthisis according to traditional homeopathic practice. Indications: According to Enderlein, Aspergillus niger belongs to the microbial cycle of the tuberculosis...
Drug Facts acc. to Enderlein Symbio Muc 4X Drops (10ml) (NDC 69710-135-11) Active Ingredient PurposeMucor racemosus 4X.....................Internal indications* Uses This homeopathic remedy is indicated for symptoms associated with circulatory and congestive conditions according to traditional homeopathic practice. * These statements are based upon traditional homeopathic practice. They...